Pharmaceutical Research: How Drugs Like Daclatasvir Work
Did you know a single molecule can stop a virus from replicating? That simple idea drives much of pharmaceutical research. On this page I explain how researchers find targets, test compounds, and move promising drugs into patients—using daclatasvir as a clear example.
Pharmaceutical research covers everything from basic lab work to large clinical trials. It starts with an idea: identify a viral protein, an enzyme, or a receptor you can interfere with. Then scientists test dozens or hundreds of compounds in cells and animals to find ones that work and seem safe. The most promising candidates go into human trials where safety and effectiveness are measured step by step.
How antiviral drugs are studied
Researchers usually follow this path: target identification, preclinical testing, and clinical trials (Phase I to III). In preclinical tests they check if a compound blocks the virus in cell cultures and whether it causes harm in animals. Phase I focuses on safety in a small group of healthy volunteers. Phase II looks for early signs of effectiveness and the right dosage. Phase III compares the drug to current treatments in larger groups to confirm benefit and monitor side effects. Regulators then review the data before approving the drug for wider use.
Trials measure clear outcomes: viral load drop, cure rates, symptom improvement, and safety markers. That’s why you’ll often see numbers like “sustained virologic response” in hepatitis C studies—that’s a concrete endpoint showing the virus stays undetectable after treatment. Real-world studies after approval keep tracking rare side effects or how the drug works in more diverse patients.
Daclatasvir — a practical example
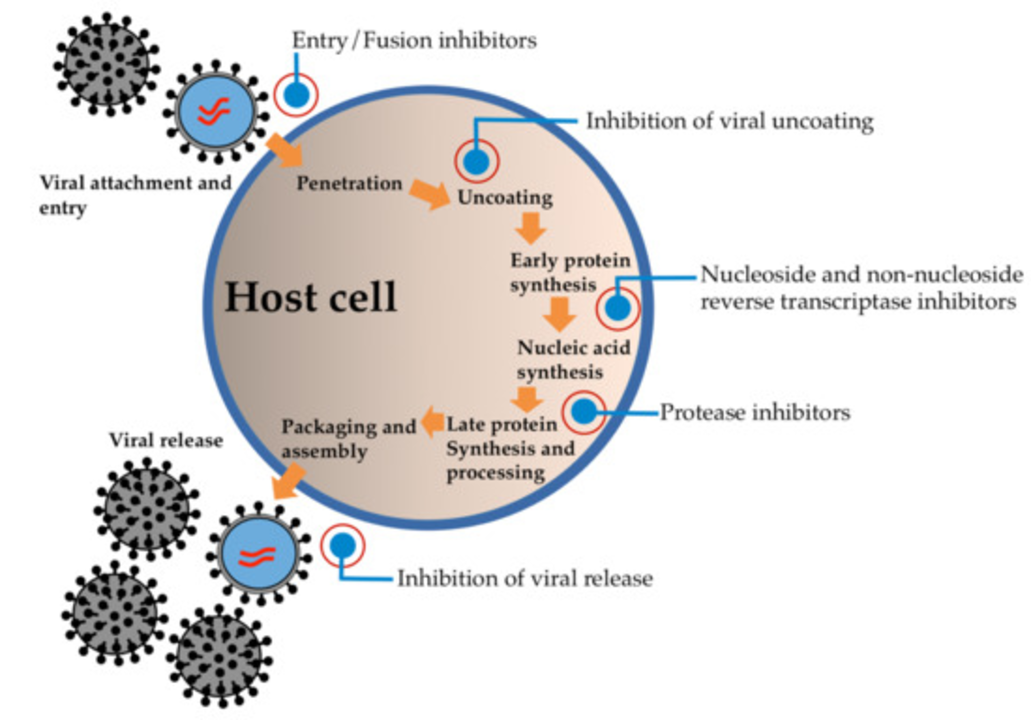
Daclatasvir targets a hepatitis C protein called NS5A. NS5A helps the virus copy and package its genome. Daclatasvir binds to NS5A and blocks those steps, which shuts down replication. When combined with other antivirals that hit different viral targets, daclatasvir helps achieve cure rates measured as undetectable virus weeks after treatment ends.
Our post "The Science Behind Daclatasvir: How Does It Work?" breaks this down with simple diagrams and data from trials so you can see why the drug changed Hep C treatment. It also covers resistance: viruses can mutate NS5A, so researchers track which combinations keep the virus suppressed and which patient groups need adjusted doses.
Want to read research yourself? Check the trial size, whether the study was randomized and double-blind, the exact endpoints, and who funded the work. That tells you a lot about how reliable the findings are. If you find a promising study, talk with a clinician before making health choices—research is useful, but individual care needs a doctor’s judgment.
If you’re curious about other drug mechanisms or want plain-language summaries of recent studies, browse our articles. I aim to explain the science clearly so you can understand how drugs are developed and what the results actually mean for patients.
When Do Drug Patents Expire? Understanding the 20-Year Term and Real-World Timelines
Drug patents last 20 years from filing, but most drugs lose exclusivity in just 7-12 years due to long development times. Learn how patent extensions, regulatory exclusivity, and legal tactics shape when generics enter the market.
Read MoreSecondary Patents: How Pharmaceutical Brands Extend Market Exclusivity
Secondary patents let drug companies extend exclusivity by patenting minor changes to existing medicines-delaying generics and keeping prices high. Learn how they work, who benefits, and why they’re under scrutiny.
Read MoreForeign Manufacturing of Generics: How the FDA Oversees Drug Quality Abroad
The FDA has ended its double standard in inspecting foreign generic drug factories. Unannounced inspections are now the norm, forcing global manufacturers to meet the same quality standards as U.S. plants. Here’s how it works-and what it means for your medications.
Read MoreStatistical Analysis in BE Studies: How to Calculate Power and Sample Size Correctly
Learn how to correctly calculate power and sample size for bioequivalence studies to meet FDA and EMA standards. Avoid common pitfalls that lead to study failure.
Read MoreHow to Use the FDA Orange Book and Purple Book for Drug Safety Information
Learn how to use the FDA's Orange Book and Purple Book to verify drug safety, spot withdrawn medications, and choose safe biosimilars and generics. Essential for pharmacists, providers, and informed patients.
Read MoreFDA Bioequivalence Standards for NTI Drugs: What You Need to Know
The FDA enforces stricter bioequivalence standards for narrow therapeutic index (NTI) drugs like warfarin, phenytoin, and digoxin to prevent dangerous dosing variations. Learn how these rules differ from standard generics and why they matter for patient safety.
Read MoreBridging Studies for NTI Generics: Ensuring Safety and Efficacy
NTI generics require stricter bioequivalence studies than standard generics to ensure safety. Learn how bridging studies, FDA guidelines, and complex trial designs protect patients using critical medications like warfarin and levothyroxine.
Read MorePharmacogenomics Testing: How Your Genes Decide Which Medications Work for You
Pharmacogenomics testing uses your DNA to predict how you'll respond to medications, reducing side effects and improving outcomes-especially for antidepressants, heart drugs, and painkillers. It's not science fiction-it's here.
Read MoreA Deep Dive into the Pharmacokinetics of Bisoprolol
Bisoprolol is a widely used beta-blocker with unique pharmacokinetics that make it safer and more predictable than older options. Learn how it’s absorbed, metabolized, and cleared - and why it’s often the preferred choice for patients with kidney or liver issues.
Read MoreThe Science Behind Daclatasvir: How Does It Work?
As a blogger, I recently delved into the fascinating world of Daclatasvir, a highly effective antiviral medication used to treat Hepatitis C. The science behind this wonder drug lies in its ability to block the action of a specific protein called NS5A, which is crucial for the virus to replicate. By doing so, Daclatasvir disrupts the life cycle of the Hepatitis C virus and prevents it from multiplying. This, in turn, helps the body's immune system to clear the virus, ultimately curing the infection. It's truly amazing to see how this simple yet ingenious mechanism has revolutionized the treatment of Hepatitis C, offering a new ray of hope to millions of patients worldwide.
Read More